Are you struggling with peak tailing in your HPLC results? You’re not alone.
Peak tailing can make your chromatograms look messy, reduce the accuracy of your data, and shake your confidence in the analysis. But the good news is, peak tailing is usually fixable once you know what’s causing it. You’ll discover simple, practical steps to identify and troubleshoot peak tailing problems in your HPLC system.
Whether it’s a chemical interaction, a column issue, or a system setup problem, you’ll learn how to tackle it effectively and get sharper, more reliable peaks every time. Keep reading to master peak tailing troubleshooting and improve your HPLC performance today.

Credit: www.youtube.com
Causes Of Peak Tailing
Peak tailing in HPLC affects data quality and accuracy. It makes peaks look stretched and uneven, complicating analysis. Understanding what causes peak tailing helps improve your results. Causes generally fall into two groups: chemical interactions and physical issues. Each plays a key role in peak shape and performance.
Chemical Interactions
Chemical interactions occur between the sample and the column surface. Acid-base reactions often cause tailing. For example, residual silanol groups on silica columns can interact with basic analytes. This interaction slows down the elution, creating tailing peaks.
Strong sample solvents can disrupt the balance in the mobile phase. This leads to uneven peak shapes. Mobile phase pH also affects these interactions. If the pH is not suitable, analytes may bind too strongly to the column.
Contaminants in the sample or mobile phase can cause secondary interactions. These contaminants may stick to the column and trap analytes briefly. This trapping leads to asymmetric peaks and poor resolution.
Physical Issues
Physical problems in the HPLC system also cause peak tailing. Column voids or blockages can disturb the flow path. Uneven flow causes some analyte molecules to travel slower, stretching the peak.
Dead volume, or extra space in the system, dilutes the sample band. This dilution makes peaks broader and tailing. Poor column packing or damage leads to irregular flow patterns inside the column.
Improper system setup, such as loose fittings or worn tubing, affects pressure stability. Pressure changes cause inconsistent flow, resulting in peak tailing. Regular maintenance and correct assembly reduce these physical problems.
Credit: uhplcs.com
Chemical Factors
Chemical factors significantly influence peak tailing in HPLC analysis. These factors affect how analytes interact with the stationary phase and mobile phase. Understanding and controlling chemical interactions can reduce peak tailing and improve chromatographic results.
Acid-base Interactions
Acid-base interactions cause peak tailing by affecting analyte ionization. Changes in pH alter the charge of analytes and stationary phase sites. This leads to strong interactions that slow down elution. Controlling mobile phase pH reduces these unwanted interactions. Using buffers stabilizes pH and minimizes peak tailing.
Sample Solvent Strength
Sample solvent strength influences how analytes enter the column. Strong solvents can cause peak tailing by disrupting interactions with the stationary phase. Using a sample solvent similar to the mobile phase prevents peak distortion. Diluting samples in weaker solvents also helps achieve sharper peaks.
Active Sites On Columns
Active sites on the column surface cause peak tailing by binding analytes. Residual silanol groups on silica columns often create these sites. These interactions delay analyte elution and cause asymmetric peaks. Using end-capped or base-deactivated columns reduces active site effects. Proper column maintenance also limits active site exposure.
Analyte And Metal Interactions
Metal ions in the system react with analytes, causing peak tailing. Metals leached from plumbing or column hardware form complexes with analytes. These complexes stick to the stationary phase longer than expected. Adding metal chelators or using metal-free components limits these interactions. Regular system cleaning prevents metal buildup and improves peak shape.
Physical Factors
Physical factors often cause peak tailing in HPLC. These issues affect the column’s performance and the quality of chromatograms. Identifying and fixing these physical problems improves peak shape and accuracy. Below are some common physical factors that lead to peak tailing.
Column Blockage And Contamination
Blockages in the column reduce flow and cause uneven peak shapes. Contaminants build up inside the column, trapping analytes and causing tailing. Regular cleaning or replacing the column can clear blockages. Using filtered and clean samples prevents contamination and maintains column health.
Dead Volume Effects
Dead volume means extra space where the sample can spread out. This spreading causes peak broadening and tailing. It happens at connections, tubing, or poorly matched parts. Keeping dead volume to a minimum sharpens peaks and improves resolution.
Column Voids And Degradation
Voids are gaps inside the packed column material. They cause uneven flow paths and peak tailing. Over time, column material can degrade, leading to void formation. Repacking or replacing the column restores uniform flow and reduces tailing.
Mass Overload
Mass overload happens when too much sample is injected. The column cannot separate all components properly. This leads to tailing peaks and poor resolution. Using smaller sample amounts keeps peaks sharp and well-defined.
Mobile Phase Optimization
Optimizing the mobile phase is crucial to reduce peak tailing in HPLC. The mobile phase directly affects the interaction between analytes and the column. Proper mobile phase composition improves peak shape and resolution. Adjusting buffers, pH, solvents, and additives can minimize tailing and enhance results.
Buffer Selection And Ph Control
Choosing the right buffer stabilizes the mobile phase pH. Buffer pH affects analyte ionization and interaction with the stationary phase. A stable pH reduces secondary interactions causing tailing. Use buffers with suitable pKa values near the target pH. Consistent pH control keeps peak shapes sharp and symmetrical.
Use Of Ion-pairing Agents
Ion-pairing agents help control interactions between charged analytes and the column. They form neutral complexes with ions, reducing tailing caused by strong ionic interactions. Select ion-pair reagents compatible with your analytes and column chemistry. Proper concentration of ion-pairing agents improves peak symmetry and retention time consistency.
Choosing Appropriate Solvents
The solvent type affects analyte solubility and retention behavior. Use solvents that dissolve all sample components well. Mixing polar and non-polar solvents changes elution strength and peak shape. Avoid solvents that cause excessive analyte-column interaction. Adjust solvent ratios to find the best balance for sharp peaks and minimal tailing.
Column Selection And Maintenance
Proper column selection and maintenance play a key role in reducing peak tailing in HPLC. Choosing the right column helps minimize unwanted interactions between the analyte and the stationary phase. Regular maintenance ensures the column performs optimally, providing sharp, symmetrical peaks. Neglecting these steps often leads to poor peak shapes and inaccurate results.
Base-deactivated And End-capped Columns
Base-deactivated columns have fewer active sites that cause tailing. They reduce interactions with polar analytes, improving peak shape. End-capped columns have silanol groups blocked to prevent unwanted bonding. These columns work well for acidic and basic compounds. Using them can lower peak tailing and improve reproducibility.
Guard Columns And Filters
Guard columns protect the main column from dirt and particles. They catch contaminants that cause blockages and peak distortion. Filters remove impurities from the mobile phase and sample. Regular use of guard columns and filters extends column life. They also help maintain consistent peak shapes during analysis.
Column Flushing And Replacement
Flushing columns regularly removes trapped contaminants and buildup. It restores column performance and reduces tailing. Use appropriate solvents for flushing based on your sample and mobile phase. Replace columns when flushing no longer improves peak shape. Timely replacement avoids long analysis times and poor data quality.
Instrumental Adjustments
Instrumental adjustments play a vital role in resolving peak tailing issues in HPLC. These tweaks focus on the hardware side to improve peak shape and accuracy. Correct adjustments ensure smoother flow paths and reduce unwanted interactions inside the system. Small changes can lead to clearer, sharper peaks and better data quality.
Reducing Dead Volume
Dead volume refers to extra space inside the HPLC system where the sample can spread out. This spreading causes peak tailing and loss of resolution. Use shorter columns and connect parts with minimal internal space. Avoid using unnecessary adapters or long tubing. Keeping dead volume low helps maintain sharp and symmetrical peaks.
Injector Volume Optimization
The volume injected into the system affects peak shape. Large injection volumes can cause peak tailing by overloading the column. Adjust the injector to deliver smaller, consistent volumes. Use autosamplers with precise control to reduce variability. Proper injection volume keeps peaks tight and well-defined.
Tubing And Fitting Checks
Loose or damaged tubing and fittings create leaks and extra dead volume. These problems lead to poor peak shapes and unreliable results. Inspect all connections regularly. Tighten fittings and replace worn tubing immediately. Using high-quality, compatible tubing reduces risk of leaks and peak distortion.
Sample Preparation Tips
Proper sample preparation is key to reducing peak tailing in HPLC analysis. The way you prepare your sample affects the interaction with the column and the shape of the peaks. Careful attention to diluent choice, sample load, and filtration can improve peak symmetry. These steps minimize unwanted interactions and help produce clear, sharp peaks.
Diluent Selection
Use a diluent similar to the mobile phase. This prevents strong solvent effects that cause peak tailing. Avoid solvents that interact with the stationary phase or analyte. A proper diluent keeps the sample stable and compatible with the column.
Sample Load Management
Do not overload the column with too much sample. Excess sample can saturate active sites and cause tailing. Use the smallest amount needed for detection. Diluting highly concentrated samples helps maintain peak shape and resolution.
Filtration Techniques
Filter samples before injection to remove particulates. Particles can block the column and cause irregular flow. Use a 0.22 or 0.45-micron filter for best results. Clean samples reduce noise and maintain consistent peak shapes.
Troubleshooting Workflow
Peak tailing in HPLC affects the accuracy and clarity of your results. A clear troubleshooting workflow helps identify and fix the cause step-by-step. This method saves time and improves the quality of your chromatograms. Follow a structured process to find the root of the problem and apply the right solution.
Benchmarking With Known Methods
Start by comparing your results with trusted, established methods. Use a standard sample and a well-known protocol. This helps confirm if the issue is with your system or your method. If the peak tailing appears in the benchmark test, the problem likely lies in the instrument or column. If not, review your sample preparation or mobile phase.
Isolating The Problem Source
Next, isolate each part of your HPLC system. Check the column, mobile phase, injector, and detector one by one. Swap components with fresh or tested ones to find the faulty part. Inspect the column for damage or contamination. Verify mobile phase pH and composition. Clean or replace parts as needed to remove sources of peak tailing.
Systematic Testing
Perform step-by-step tests after each change. Run blank injections to check for system contamination. Adjust mobile phase pH and buffer strength systematically. Test different flow rates and temperatures. Document each change and its effect on peak shape. This careful testing helps pinpoint the exact cause and ensures the best correction.
Preventive Practices
Preventive practices help avoid peak tailing issues in HPLC analysis. They keep the system stable and reliable. Following these steps ensures better peak shapes and accurate results. Regular care of the column, instrument upkeep, and method checks all play vital roles. These actions reduce downtime and improve sample quality.
Regular Column Care
Column health is essential for sharp peaks. Rinse the column with appropriate solvents after each run. Avoid using harsh chemicals that damage the stationary phase. Store the column properly to prevent drying or contamination. Monitor column pressure and replace it when signs of wear appear. Clean columns regularly to remove blockages and buildup.
Routine Instrument Maintenance
Keep the HPLC instrument in good condition to prevent tailing. Check and clean the injector, tubing, and detector frequently. Replace worn seals and filters to avoid leaks and contamination. Calibrate pumps and detectors to maintain consistent flow and detection. Regular maintenance reduces instrument-related peak distortion and errors.
Consistent Method Validation
Validate methods periodically to confirm accuracy and precision. Test new batches of solvents and reagents before use. Adjust mobile phase composition to optimize peak shape. Verify system suitability parameters like retention time and tailing factor. Consistent validation helps catch potential problems early and keeps results reliable.
Evaluating Peak Quality
Evaluating peak quality is essential in troubleshooting peak tailing in HPLC. Good peak quality ensures accurate identification and quantification of compounds. Poor peak shapes can indicate problems with the system or the method. Focus on key parameters helps identify the cause of tailing. Assessing these factors improves the reliability of your results.
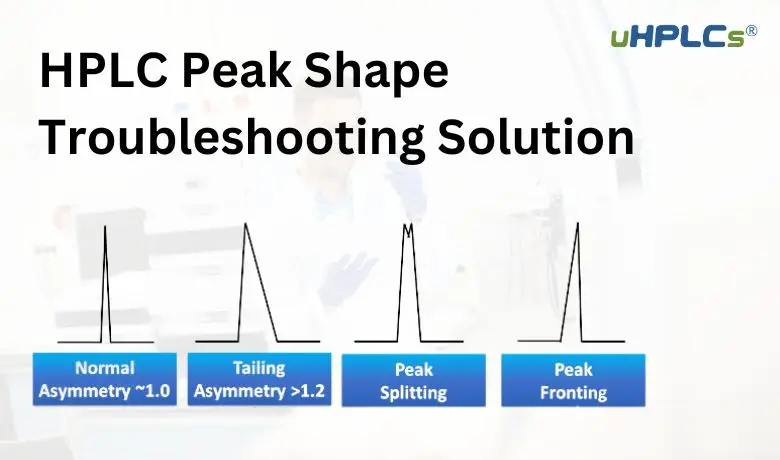
Tailing Factor Criteria
The tailing factor measures how symmetrical a peak is. It compares the front and tail widths at 5% of peak height. A tailing factor close to 1 means a symmetrical peak. Values above 1.5 indicate significant tailing. Consistent tailing factors suggest stable system performance. Check this factor for every critical peak in your analysis.
Impact On Quantification
Tailing peaks affect the accuracy of quantification. They distort peak area and height measurements. This distortion leads to incorrect concentration calculations. Severe tailing can cause overlapping peaks and poor resolution. Quantitative results become unreliable without proper peak shape. Regular monitoring reduces errors in data interpretation.
Assessing Peak Purity
Peak purity confirms that a peak represents a single compound. Impure peaks may cause tailing or fronting. Use spectral analysis or diode array detection to check purity. Impurities or co-eluting substances affect peak symmetry. Confirming purity helps avoid misidentification of compounds. Pure peaks increase confidence in your HPLC results.

Credit: www.youtube.com
Frequently Asked Questions
What Can Cause Peak Tailing In Hplc?
Peak tailing in HPLC occurs due to analyte interactions with active column sites, acidic silanol groups, or strong sample solvents. Contamination, column blockage, and improper mobile phase pH also cause tailing. Use buffered mobile phases, base-deactivated columns, and proper sample preparation to reduce peak tailing effectively.
Why Is Peak Tailing A Problem?
Peak tailing distorts chromatograms, lowers resolution, and impairs accurate quantification. It reduces confidence in analytical results and complicates data interpretation.
How To Fix Tailing In Chromatography?
Fix tailing by optimizing mobile phase pH and buffer, using base-deactivated columns, filtering samples, and reducing strong solvents. Flush or replace contaminated columns.
What Is An Acceptable Tailing Factor?
An acceptable tailing factor typically ranges from 0. 9 to 1. 5. Values above 2 indicate poor peak shape and require troubleshooting.
What Causes Peak Tailing In Hplc?
Peak tailing occurs due to interactions between analytes and active sites in the column. It often results from secondary chemical interactions or column contamination. Improper mobile phase pH or sample solvent strength can also cause tailing.
Conclusion
Peak tailing in HPLC can affect your results and analysis quality. Simple changes often improve peak shape and accuracy. Adjusting mobile phase pH and using buffers helps reduce tailing. Choosing the right column and sample solvent also matters. Regular maintenance and cleaning keep the system running smoothly.
Keep testing small adjustments to find the best settings. With patience, tailing issues become manageable and less frustrating. Consistent troubleshooting leads to clearer, sharper chromatograms every time.




